Open access events exploring the current understanding of the field and visions of the emerging practices along with the translational implications.
By Shraddha Thakkar, Ph.D.; Beata Sweryda-Krawiec, Ph.D.; Rakesh Gollen, Ph.D.; and Rajesh Patil, Ph.D.

The tailoring of therapies and the design of personalized medicines for individuals or subpopulations of individuals based on their unique genetic makeup or other specific biomarkers is the main promise behind precision medicine—also variously known as individualized medicine, personalized medicine, or genomic medicine. Recent advances in the field of diagnostics, biomarkers, and genomics have made personalized medicine a reality. The ultimate objective is to give the right medicine to the right patient at the right time, thus maximizing the benefits to patients with the potential to reduce the cost to society by focusing treatment on the individuals most likely to benefit. There is clear evidence that personalized medicine is gaining momentum. Examples include, but are not limited to, Warfarin (small molecule—anticoagulant), Herceptin (biologic—cancer monoclonal antibody), and the more recently approved Provenge (vaccine for prostate cancer).
Success is challenging because it requires the translation of pharmacogenomics (PGX) and pharmacology from the bench into the clinic. It also requires the involvement of multiple players to understand and implement variations in drug metabolism to avoid adverse events and improve efficacy. Thus, advancing personalized medicine depends on numerous players: academic researchers, pharmaceutical and biotechnology companies, diagnostic companies, clinicians, information technology managers, health care providers, laboratories, policymakers, and payers. Considering the multifaceted nature of this field, the goal of the webinar series is to touch upon the crucial aspect of personalized medicine and to provide an introduction and an opportunity to explore the field. Both scientists seeking a better understanding and experts working in disease areas such as cancer, neurological disorders, and pain will benefit.
The webinar series will introduce personalized medicine and expand to provide specific details of its applications in different therapeutic areas. The series starts with an introduction to PGX to update the audience on the basics of polymorphisms, with a focus on emerging PGX practices and application. Polymorphisms in genes encoding transporters and enzymes as well as therapeutic targets have become a major clinically relevant issue in the identification of therapeutic targets and defining individuals at risk for unwanted side effects. PGX of transporters has become increasingly important as genetic variability of SLCO1B1 has been linked to response to statins and the development of myopathy. The cytochrome P450 enzymes such as CYP2D6, CYP2C9, CYP2C19, and CYP3A4 are the best-studied polymorphic enzymes associated with alterations in drug action. Thus, this webinar series will provide insights on the introduction as well as current understanding of the field but also provide visions on the emerging practices along with the translational aspect of personalized medicine. Topics covered include the following:
Technologies
The Food and Drug Administration (FDA) has evolved the regulatory process in response to the development in the various fields associated with personalized medicine. These developments have been critical in the approval of multiple therapies and diagnostics. FDA played a significant role in bridging the gap in the translation of genomics technology to clinical practice by advancing the tools necessary for evaluation of targeted therapies. The advancements of incorporating PGX-based practices go hand in hand with the advancements in the PGX technologies. To discuss the importance of having a reliable and reproducible method for measuring genetic variability, presenters will focus on the FDA-led consortium that brought together various platforms of technology and developed the gold standard for operating procedures for using genomic technology to get reliable, reproducible results. Current established technologies that help in PGX decision making will be presented.
Neurological Disease
Personalizing treatment for neurological diseases has proven difficult. The nature of therapies includes narrow therapeutic indexes, severe and potential life-threatening toxicities, and variable response rates and efficacies. The current therapeutic landscape of neurological diseases and the prospects and key hurdles for development of new therapeutics in several areas of disease with focus on personalized medicine in neurological diseases will be discussed. Areas to be covered include Alzheimer, Parkinson, frontotemporal dementias, and amyotrophic lateral sclerosis; excitability-related disorders such as epilepsy and pain; and neuropsychiatric disorders including schizophrenia, autism, depression, and bipolar disorders.
Cancer
Implementing personalized medication in cancer treatment may offer choices to anticipate differences in drug response, resistance, efficacy, and toxicity within chemotherapeutic agents and targeted immune biologic agents. This can be used to optimize treatment regimens based on patient variability. Many of the cancer treatment agents are biologics, targeting specific antigens expressed on cancer cells or blocking stimulators and signal transduction pathways of tumor growth or enhancing anticancer immune responses.
Pain
Personalized treatment is crucial for effective pain management. Both pain and treatment response are associated with large inter-individual variability. The prediction of personalized treatment regimens is further complicated by the multifactorial nature of pain. Pain is dependent on a clinical, psychosocial, neurophysiological, and biochemical component. Speakers will focus on systems pharmacology approaches for personalized treatment of pain.
Biomarkers
Another crucial area is biomarkers, which have been used in drug development and clinical practice for many years. However, there has been confusion about the definitions and inconsistent use of terminologies. Recently, an FDA-National Institutes of Health Biomarker Working Group developed a glossary of terms and definitions to ensure consistency and clarity, termed BEST (Biomarkers, EndpointS, and other Tools). Speakers will focus on the biomarker and its application in the targeted therapies.
FDA Role
In FDA’s Center for Drug Development and Research (CDER), the Genomics and Targeted Therapy Group in the Office of Clinical Pharmacology plays a crucial role for precision medicine advancements through discovery, development, and regulation. The responsibility of this group is to ensure that strategies for the targeted development and PGX are endorsed throughout all stages of drug development. Presenters will focus on the role of CDER’s Genomics and Targeted Therapy Group in maximizing the impact of individualized therapy.
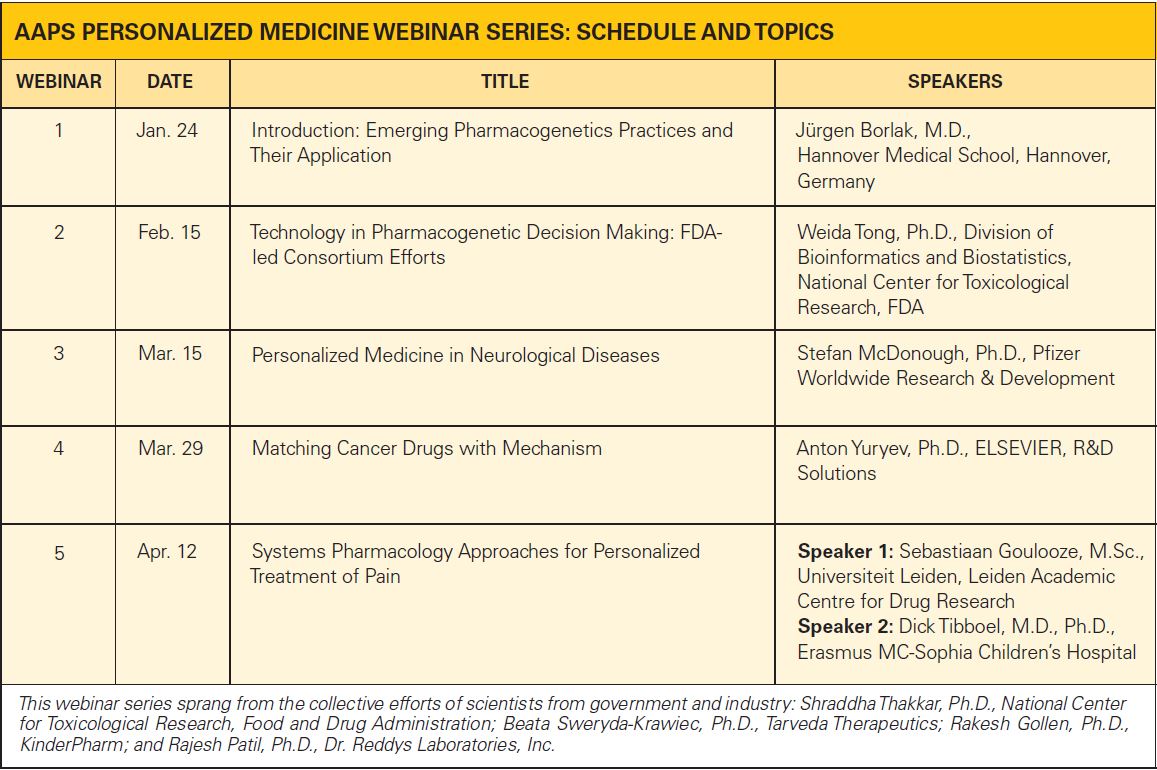
This webinar series aims to provide in-depth understanding of the field by incorporating a wide variety of experts and topics. Find details on the schedule and topics below.
Visit the Web Series to see more information and register today!