Recent Pharmaceutical Research theme delivers five expert reviews and eight research articles in this growing field.
By Wolfgang Sadee
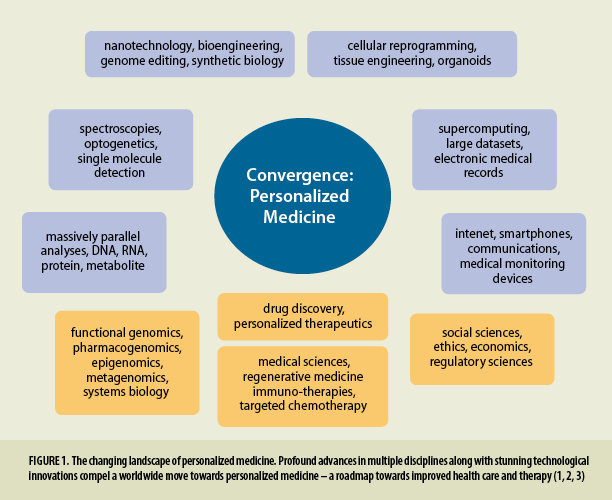
This Theme Issue of Pharmaceutical Research assembles a series of original papers and reviews on personalized therapeutics and pharmacogenomics, a rapidly expanding field fostering fundamental changes in personalized health care. Stunning advances in novel technologies and disciplines propel unprecedented insights in biomedical sciences, stimulating profound changes in global health care (1, 2, 3) and converging on personalized health care (Fig. 1). Our growing ability to gather immense information on an individual’s health status and genomic profile compels a shift of health care from one-drug-treats-all towards personalized medicine. Assembled in electronic health records, and increasingly available across health care systems, personal genomics holds promise for driving innovations in modern health care (3). We expect to gain a better understanding on how each individual’s unique characteristics reveal resilience in health, susceptibility to disease, and response to interventions and therapy.
With this background, we consider “personalized therapeutics” germane to the pharmaceutical sciences. As a result of the dramatic expansion of genomics, drug therapy is further transformed by pharmacogenomics – the use of genomics information to optimize an individual’s health care and therapy. Today genomics is understood to encompass all –omics areas, including integration between diverse data on DNA, RNA, protein, metabolite, epigenome, metagenome, etc. The articles in this issue address the confluence between therapeutics and our new understanding of the relevance of inter-individual differences, including genomics, highlighting the challenges we face and opportunities we can pursue.
With dramatic human population growth, drastic environmental changes, and rapid genetic adaptation emerging in parallel (4,5), we face growing challenges in maintaining health, preventing disease progression, and treating disease effectively. Dense cohabitation facilitates spread of infectious disease, thereby leading to an evolutionary process that strengthens the innate immune system as a primary defense mechanism. In turn, a hyper-active innate immune system results in autoimmune and inflammatory diseases that have become increasingly prevalent (6). Rapid population growth accelerates adaptations of the human genome to changing environments, favoring genetic variants that promote reproductive success but do not necessarily protect against chronic disease. Given the diversity in genetics, environment, behaviors, and social factors, drug therapy often fails to provide benefit and even causes substantial harm. Adverse drug effects, defined by WHO definition to be unavoidable even if the drug is given according to standard recommendations, are considered a leading cause of morbidity and mortality (7).
It has become evident that the maintenance of health, effective disease prevention, and therapy depend on key factors that dynamically interact with each other: environment, diet, exercise, age, sex, socioeconomic status, cultural context, and access to health care. Drug therapy is only one part of the health care continuum, and by itself unlikely to be effective unless well integrated into an individual’s personal life style. Diet intervention, exercise programs, and behavioral changes are all critical and often can replace drug therapy. For example, rigorous exercise might not only prevent disease and slow aging, but could also replace drug therapy under certain circumstances (8). Similarly, near universal use of smart phone devices leads to new health apps that monitor an individual’s physical status and can lead to life style changes that also could obviate the need for drug therapy (9). In addition, continuous measurements of symptoms, such as blood pressure and insulin, educate drug dosage schedules. This raises questions whether and when sustained release drug formulations are indeed warranted or may be counterproductive. Glucose levels and blood pressure fluctuate, often synchronous with the circadian rhythm – now recognized as an important factor in health and therapy response (10). Recently, dosage regimen adjusted to continuous personal health measures have become feasible, leading to improved health care. Moreover, genetics and genomics can serve as an additional guidepost, in a growing number of instances already part of standard care – an endeavor still in infancy but making substantive progress.
Spectacular advances in genomic medicine have led to discovery of single genes or mutations linked to the disease process. These include driver mutations in cancer amenable to targeted chemotherapy, and CFTR mutations causing protein defects that can be corrected with suitable ligands to restore normal functions. Over the past 4–5 years, ~20% of all newly FDA-approved drugs require a companion biomarker test so that only those who benefit receive the treatment (11). Some project obligatory biomarker tests to account for up to 70% of New Drug Approvals, as blockbuster drugs treating a complex disorder broadly have proven increasingly difficult to develop. We can further anticipate gene therapy (example (12)), in vivo gene editing, including the use of CRISPR (13), and immune modulation (14,15) to assume a critical role in medicine. While epigenomics focuses on chromatin and gDNA modifications that can be long lasting with profound effects on disease and drug response, a nexus between genetic variants and epigenetic marks (16) is often overlooked but highlights genetic predisposition to the influence of environmental factors. Lastly, somatic mutations that accumulate over time are thought to contribute to aging and cancer, but also could be critical to other diseases (17). If somatic mutations lead to clonal expansion, a majority of target cells could carry the mutations. For example, relatively frequent somatic mutations of TET2, which modulates the immune system by oxidizing methylated CpG, lead to clonal expansion of hematopoetic stem cells without causing cancer. In a mouse model, tet2−/− macrophages secrete more IL1b upon stimulation and promote atherosclerotic cardiovascular disease, offering new opportunities for therapies (18). Yet, common complex disorders thus far have largely defied attempts to develop clinically compelling biomarker tests even while genetic factors (broad sense heritability) contribute a substantial portion to disease risk and treatment outcome. Managing complex diseases and health maintenance into advanced age requires integration of life-style changes and dietary programs, with drug therapies as complementary strategies. Similar developments in individualized therapies are beginning to emerge in veterinary medicine (19), and human therapeutics can benefit from those advances that can be more readily implemented in animals.
The articles in this Theme Issue critically evaluate the influence of these interacting factors, the search for critical genetic variants, and evaluation of clinical utility, in an effort to set beacons for therapeutic advances to come. Taken together, these articles provide a topical cross section highlighting developments in personalized therapeutics and pharmacogenomics.
This article was reproduced with permission from Springer Nature. The original, full-text article can be found online.
References
- Zusi K. Stunning advances over the past 30 years. The Scientist. 2016;10:32–44.
- Sharp P, Jacks T, Hockfield S. Capitalizing on convergence for health care. Science. 2016;352(6293):1522–3. doi:10.1126/science.aag2350.
- Denny JC. Translational Bioinformatics: Big data in EHRs, Omics for drugs, and personal Genomics. Nashville: Vanderbilt University Medical Center, IMIA Yearbook of Medical Informatics; 2014: 199–205.
- Fan S, Hansen ME, Lo Y, Tishkoff SA. Going global by adapting local: review of recent human adaptation. Science. 2016;354:54–9. doi:10.1126/science.aaf5098.
- Gluckman P, Beedle A, Hanson M. Principles of evolutionary medicine. Oxford: Oxford Biology; 2009.
- Azad A, Sadee W, Schlesinger LS. Innate immune gene polymorphisms in tuberculosis. Infect Immun. 2012;80:3343–59. doi:10.1128/IAI.00443-12.
- Phillips KA, Veenstra DL, Oren E, Lee K, Sadee W. Potential role of pharmacogenomics in reducing adverse drug reactions: a systematic review. J Am Med Assoc. 2001;286:2270–9. doi:10.1001/jama.286.18.2270.
- Oaklander M. The Exercise Cure. Time Magazine. 2016. Sept.12–19.
- Kvedar J. Is chronic disease management beyond our reach? The Internet of Healthy Things. 2016. http://theinternetofhealthythings.com.
- Turek FW. Circadian clocks: not your grandfather’s clock. Science. 2016;354:992–3. doi:10.1126/science .aal2613
- Personalized Medicine Coalition. Progress Report 17, no 3, May/June 2016.
- Ribeil JA, Hacein-Bey-Abina S, Payen E, et al. Gene therapy in a patient with sickle cell disease. N Engl J Med. 2017;376:848–55. doi:10.1056/NEJMoa1609677.
- Yin H, Kauffman KJ, Anderson DG. Delivery technologies for genome editing. Nat Rev Drug Discov. 2017; 16(6):387–399. doi:10.1038/nrd.2016.280.
- Leslie M. Fighting autoimmunity with immune cells. Science. 2016;353:14. doi:10.1126/science.353.6294.14.
- Reardon DA, Wen PY, Wucherpfennig KW, Sampson JH. Immunomodulation for glioblastoma. Curr Opin Neurol. 2017;30(3):361–9. doi: 10.1097/WCO.0000000000000451.
- Zaghlool SB, Al-Shafai M, Al Muftah WA, et al. Mendelian inheritance of trimodal CpG methylation sites suggests distal cis-acting genetic effects. BMC Genomics. 2017;18(1):150. doi:10.1186/s13148-016-0295-1.
- Erickson RP. Recent advances in the study of somatic mosaicism and diseases other than cancer. Curr Opin Genet Dev. 2014;26:73–8. doi:10.1016/j.gde.2014.06.001.`
- Fuster JJ, MacLauchlan S, Zuriaga MA, et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science. 2017;355:842–7. doi:10.1126/science.aag1381.
- Rood J. Individualized therapies: from human to pets - and back again. The Scientist. 2016;10:70–2.