In January 2021, The AAPS Journal published the review article γ-Hydroxybutyric Acid: Pharmacokinetics, Pharmacodynamics, and Toxicology, by Melanie A. Felmlee, University of the Pacific, Bridget L. Morse, Eli Lilly and Company, and Marilyn E. Morris, University at Buffalo.
This article examines the pharmacology, therapeutic effects, and pharmacokinetics/pharmacodynamics of gamma-hydroxybutyrate (GHB). GHB is a short-chain fatty acid found endogenously in the brain. It can be used to treat narcolepsy and alcohol withdrawal, but it is more commonly known by the street name the “date rape drug.”
GHB overdoses are common because of the steep concentration-effect curve. This article provides pharmacological and toxicological examination of GHB, which may help drive new therapies to treat overdoses.
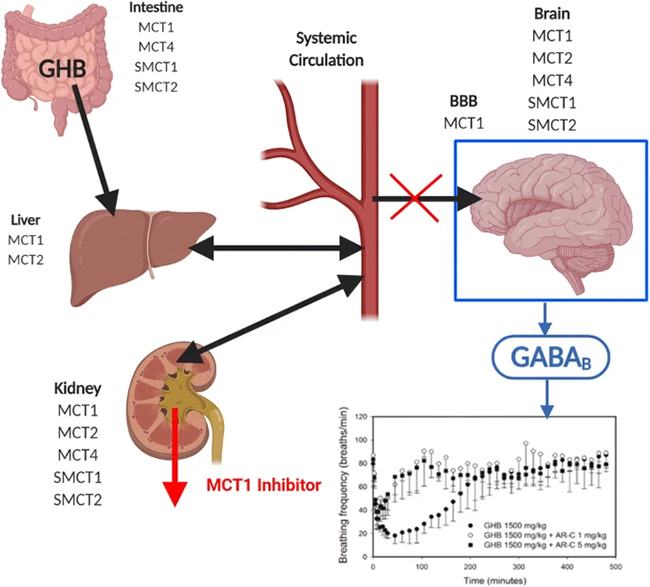
“GHB binds to both GHB and GABAB receptors in the brain, with pharmacological/toxicological effects mainly due to GABAB agonist effects. The pharmacokinetics of GHB are complex and include nonlinear absorption, metabolism, tissue uptake, and renal elimination processes. GHB is a substrate for monocarboxylate transporters, including both sodium-dependent transporters (SMCT1, 2; SLC5A8; SLC5A12) and proton-dependent transporters (MCT1–4; SLC16A1, 7, 8, and 3), which represent significant determinants of absorption, renal reabsorption, and brain and tissue uptake.”
This review article is part of The AAPS Journal theme issue Celebrating Women in the Pharmaceutical Sciences, guest edited by Diane Burgess, Marilyn Morris, and Meena Subramanyam.
Read more recent articles on pharmacokinetics and pharmacodynamics in AAPS’ three active journals. Articles are freely available to AAPS members by accessing the journals through www.aaps.org/journals.
The AAPS Journal
Population Pharmacokinetics of Intracellular 5-Fluorouridine 5′-Triphosphate and its Relationship with Hand-and-Foot Syndrome in Patients Treated with Capecitabine
By Julie M. Janssen, Bart A. W. Jacobs, Jeroen Roosendaal, Ellen J. B. Derissen, Serena Marchetti, Jos H. Beijnen, Alwin D. R. Huitema, and Thomas P. C. Dorlo
Multiscale and Translational Quantitative Systems Toxicology, Pharmacokinetic-Toxicodynamic Modeling Analysis for Assessment of Doxorubicin-Induced Cardiotoxicity
By Tanaya R. Vaidya, Hardik Mody, Yesenia L. Franco, Ashley Brown, and Sihem Ait-Oudhia
Open Access
Population Pharmacodynamic Modeling Using the Sigmoid Emax Model: Influence of Inter-individual Variability on the Steepness of the Concentration–Effect Relationship. a Simulation Study
By Johannes H. Proost, Douglas J. Eleveld, and Michel M. R. F. Struys
Open Access
Comparison of the ELISA and ECL Assay for Vedolizumab Anti-drug Antibodies: Assessing the Impact on Pharmacokinetics and Safety Outcomes of the Phase 3 GEMINI Trials
By Timothy Wyant, Lili Yang, and Maria Rosario
Pharmaceutical Research
Open Access
Application of a Two-Analyte Integrated Population Pharmacokinetic Model to Evaluate the Impact of Intrinsic and Extrinsic Factors on the Pharmacokinetics of Polatuzumab Vedotin in Patients with Non-Hodgkin Lymphoma
By Dan Lu, Tong Lu, Rong Shi, Leonid Gibiansky, Priya Agarwal, Colby S. Shemesh, Randall C. Dere, Uzor Ogbu, Jamie Hirata, Pascal Chanu, Sandhya Girish, Jin Yan Jin, Chunze Li, and Dale Miles
Open Access
Physiologically Based Pharmacokinetic Models of Probenecid and Furosemide to Predict Transporter Mediated Drug-Drug Interactions
By Hannah Britz, Nina Hanke, Mitchell E. Taub, Ting Wang, Bhagwat Prasad, Éric Fernandez, Peter Stopfer, Valerie Nock, and Thorsten Lehr
Open Access
Ocular Physiologically Based Pharmacokinetic Modeling for Ointment Formulations
By Maxime Le Merdy, Jessica Spires, Viera Lukacova, Ming-Liang Tan, Andrew Babiskin, Xiaoming Xu, Liang Zhao, and Michael B. Bolger
Evaluating Dermal Pharmacokinetics and Pharmacodymanic Effect of Soft Topical PDE4 Inhibitors: Open Flow Microperfusion and Skin Biopsies
By Stefan Eirefelt, Joanna Hummer, Line Hollesen Basse, Malene Bertelsen, Fredrik Johansson, Thomas Birngruber, Frank Sinner, Jens Larsen, Simon Feldbæk Nielsen, and Maja Lambert
Simultaneous Pharmacokinetic Analysis of Nitrate and its Reduced Metabolite, Nitrite, Following Ingestion of Inorganic Nitrate in a Mixed Patient Population
By Andrew R. Coggan, Susan B. Racette, Dakkota Thies, Linda R. Peterson, and Robert E. Stratford Jr
AAPS PharmSciTech
Preparation, Characterization, and In Vitro Pharmacodynamics and Pharmacokinetics Evaluation of PEGylated Urolithin A Liposomes
By Shengfu Yi, Cong Zhang, Junjie Hu, Yan Meng, Liang Chen, Huifan Yu, Shan Li, Guihong Wang, Guohua Zheng, and Zhenpeng Qiu
Transdermal Delivery of Salmon Calcitonin Using a Dissolving Microneedle Array: Characterization, Stability, and In vivo Pharmacodynamics
By Lu Zhang, Yingying Li, Fang Wei, Hang Liu, Yushuai Wang, Weiman Zhao, Zhiyong Dong, Tao Ma, and Qingqing Wang
Exploratory Study on Lercanidipine Hydrochloride Polymorphism: pH-Dependent Solubility Behavior and Simulation of its Impact on Pharmacokinetics
By Ilia Alekseevich Repin, Raimar Loebenberg, John DiBella, António C. L. Conceição, Manuel E. Minas da Piedade, Humberto G. Ferraz, Michele G. Issa, Nadia A. Bou-Chacra, Catharine F. M. Ermida, and Gabriel L. B. de Araujo